 
Carbon monoxide results from burning fossil fuels it is also an important industrial chemical used in manufacturing detergents. Formaldehyde (CH 2O) is a chemical that is used to preserve tissues you may be familiar with its odour from anatomy lab. To help us think about formal charges, let's look at a few small molecules that all contain carbon-oxygen multiple bonds but that are slightly different from each other. valence rules can act as flags to alert you that formal charges are present.formal charge is often present if the atom does not have its usual number of bonds.If an atom donates fewer electrons than normal and everyone still has an octet, it must be getting extra electrons from somewhere else. If an atom needs to donate more electrons than normal in order for everyone to get an octet, it will have a positive formal charge. They tell us if one atom is donating extra electrons to another to give it an octet. You will see why below.įormal charges are an important book-keeping device that we use in Lewis structures. In a Lewis structure of the compound, the carbon has a formal negative charge. However, the fact that the carbon is attracted to a metal cation begs the question: Is the carbon an anion? Yes, in a sense. Why does the molecule behave in this way? There are actually a number of reasons. atoms with negative formal charges often donate electrons.atoms with positive formal charges often attract electrons.formal charge can help us predict how a molecule behaves.In the case of hemoglobin, because the carbon monoxide binds very strongly to the iron, the CO blocks the position where oxygen would normally be bound and carbon monoxide poisoning results. 
When exposed to transition metal cations such as the iron in hemoglobin (Fe2+), the carbon is attracted to and binds to the metal. chemical reactions occur via attraction and donation of electronsįormal charge can help us to understand the behavior of carbon monoxide, CO. 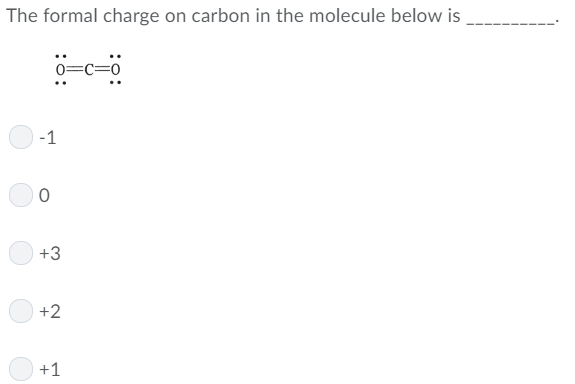
Old chemical bonds are broken when one atom takes the bonding electrons away from another atom. New chemical bonds are formed by sharing electrons. That is because reactivity has to do with the reorganization of electrons between atoms. One of the tools that we will eventually use to understand reactivity is formal charge. Looking at the structure of a molecule can help us to understand or to predict the behavior of that compound. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
